Alenura™
Millions of people worldwide suffer from acute, recurring pain flares in the bladder.
Alenura™ aims to become the first drug specifically approved to relieve pain Interstitial Cystitis / Bladder Pain Syndrome (IC/BPS)
The medical indication
Symptoms include pelvic pressure and burning discomfort, increased urinary frequency, impacting every aspect of daily life. Episodes of painful flares last hours or days: the direct impact of the indication is amplified by stress, fatigue and even depression.
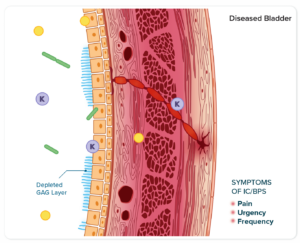
In healthy bladders, the inner lining of the bladder wall prevents urine from penetrating and reaching sensitive nerve ends.
A weakened inner layer of the bladder exposes the underlying tissue (urothelium) to toxic components in urine, like potassium.
The condition is suffered by millions worldwide, with estimates for the U.S. market alone starting at 6 million people, the majority of which are women.
|
|
U.S. patients (Low estimate – high estimate) |
| Women (1) | 3.5M – 8.6M |
| Men (2) | 2.4M – 5.3M |
| Total | 5.9M – 13.9M |
(1) RAND Study: J Urol. 2011 August, 186(2): 540–544. doi:10.1016/j.juro.2011.03.132
(2) RICE Study: J Urol. 2013 January, 189(1): 141–145. doi:10.1016/j.juro.2012.08.088
Annually, 3 million instillations of unapproved, compounded drug products take place in the US. Physicians are reimbursed for the instillation procedure but not for off label use of unapproved instillation products.
Our solution
No standardized treatment protocol for IC/BPS exists. Alenura™ aims to become the first product specifically approved for IC/BPS.
Current treatment protocols vary and have clear limitations. Alenura™ could become the first drug specifically approved to relieve pain Interstitial Cystitis / Bladder Pain Syndrome (IC/BPS), bringing reliable and standardized treatments to millions of patients.
Alenura™ aims to provide fast relief for acute painful symptoms, through a ready-to-use prefilled syringe for intra-vesicular administration through an instillation procedure.
Urologists recognize the unmet medical need and express an interest in Alenura™
We are developing a combination product of alkalinized lidocaine and heparin sodium, for which the mechanism of action is widely known thus reducing the risk for our drug development.
Alkalinized Lidocaine
- Penetrates the bladder wall
- Provides immediate pain relief
- Helps reduce inflammation
Heparin Sodium
- Augments the inner lining of the bladder (GAG layer)
- Prevents further infiltration of toxic elements in urine
- Anti-inflammatory & anti-microbial
- Stabilizes alkalinized lidocaine
Alenura™ would be the only drug product that (1) treats pain acute flares and (2) regenerates the mucous layer of the bladder for prolonged pain relief.
Our commitment
The unique combination Alenura™ has already demonstrated efficient pain relief in controlled clinical trials, offering hope for patients in need.
In June of 2023 an ambitious phase 2 program was launched, including a 4-arm study comparing the efficacy of Alenura™ to its 2 components as well as placebo. The trial will enrol a target of 120 patients across multiple sites in the United States.
Hyloris has committed to staged investments of in total maximum USD 6.7 million. This stays within our financial goals of getting a product to market within 7 years and for less than €7 million on average.
